Targeted bacteriophages may modulate microbiota-driven inflammation and influence cognitive decline through the gut–brain axis.
Recent advances in microbiome research suggest that bacteriophages may play a broader role than previously recognized. While traditionally used to combat bacterial infections, phages are increasingly viewed as ecological modulators capable of reshaping microbial communities and influencing host physiology.
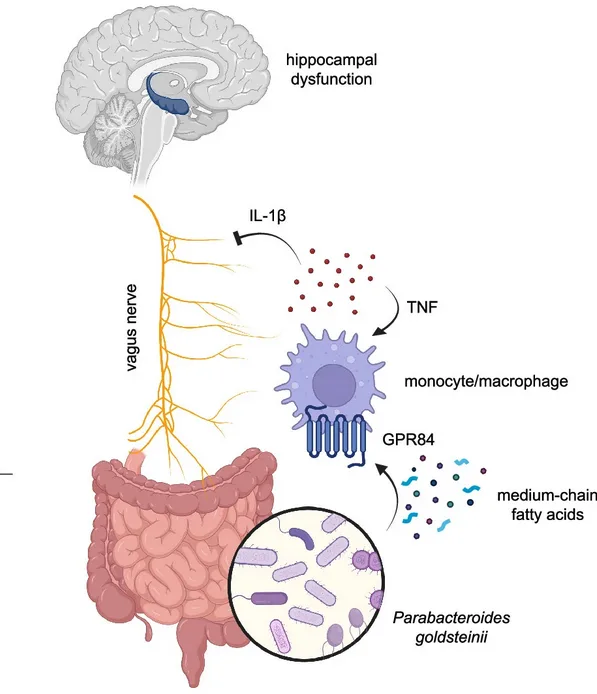
A recent Nature study led by the teams of Maayan Levy and Christoph A. Thaiss demonstrated that age-related cognitive decline may originate in the gut. The authors showed that alterations in microbiota composition trigger intestinal immune activation, disrupt vagus nerve signaling, and impair hippocampal memory. Although bacteriophages were not directly investigated, this mechanistic pathway highlights a new opportunity: phage-mediated modulation of microbiota could influence gut–brain communication and cognitive aging.
Bacteriophages naturally regulate bacterial populations and therefore can indirectly affect microbial metabolite production and inflammatory signaling. By selectively targeting specific bacteria, phages may:
• Restructure microbiota composition
• Modify production of neuroactive metabolites
• Reduce gut-derived inflammation
• Restore balanced gut–brain signaling
This ecological precision distinguishes phages from broad-spectrum approaches. Rather than eliminating entire microbial communities, phages allow targeted modulation of key bacterial drivers implicated in disease.
The implications extend beyond infection control. Phage-based strategies could potentially influence chronic inflammation, metabolic regulation, immune homeostasis, and possibly cognitive aging. By acting upstream of microbiota-driven pathways, phages may become tools for precision microbiome engineering.
Perspective
These findings support a shift in the conceptual framework of phage therapy. Instead of being viewed solely as antibacterial agents, bacteriophages may function as ecosystem regulators capable of restoring physiological communication between microbiota and host systems.
Future research will determine whether targeted phage interventions can restore gut–brain signaling and improve cognitive outcomes. If confirmed, phage therapy could evolve into a novel approach for modulating microbiota-driven processes involved in aging and neurodegeneration, expanding its therapeutic scope far beyond infectious diseases.
Reference:
Cox, T.O., Devason, A.S., de Araujo, A. et al. Intestinal interoceptive dysfunction drives age-associated cognitive decline.
Nature (2026). https://doi.org/10.1038/s41586-026-10191-6
